|
Back to Blog

According to the sigma bond rule, the number of sigma bonds in a molecule is equivalent to the number of atoms plus the number of rings minus one. Organic molecules are often cyclic compounds containing one or more rings, such as benzene, and are often made up of many sigma bonds along with pi bonds. These sigma bonds can be supplemented with other bonding interactions, such as π-back donation, as in the case of W(CO) 3( PCy 3) 2(H 2), and even δ-bonds, as in the case of chromium(II) acetate. Transition metal complexes that feature multiple bonds, such as the dihydrogen complex, have sigma bonds between the multiple bonded atoms. For example, propane is described as consisting of ten sigma bonds, one each for the two C−C bonds and one each for the eight C−H bonds. The concept of sigma bonding is extended to describe bonding interactions involving overlap of a single lobe of one orbital with a single lobe of another. Sigma Bonds the Pi Bonds are more reactive because electrons are not tightly held by the nuclei.Sigma bonds are obtained by head-on overlapping of atomic orbitals. And if we talk about the reactivity in Pi Bonds vs. Sigma Bond is free and dependent on high stability, while the Pi Bond is not free and has less stability. Pi Bonds are in their overlapping of atomic orbitals as Sigma Bond has a face to face overlapping while Pi Bond had side to side overlapping. Conclusionīoth the sigma and Pi Bonds are types of the covalent bond and very essential in determining the shape of the molecules-the main difference in sigma vs. Pi Bonds can not exist independently, as they only exist in the presence of the Sigma Bond. 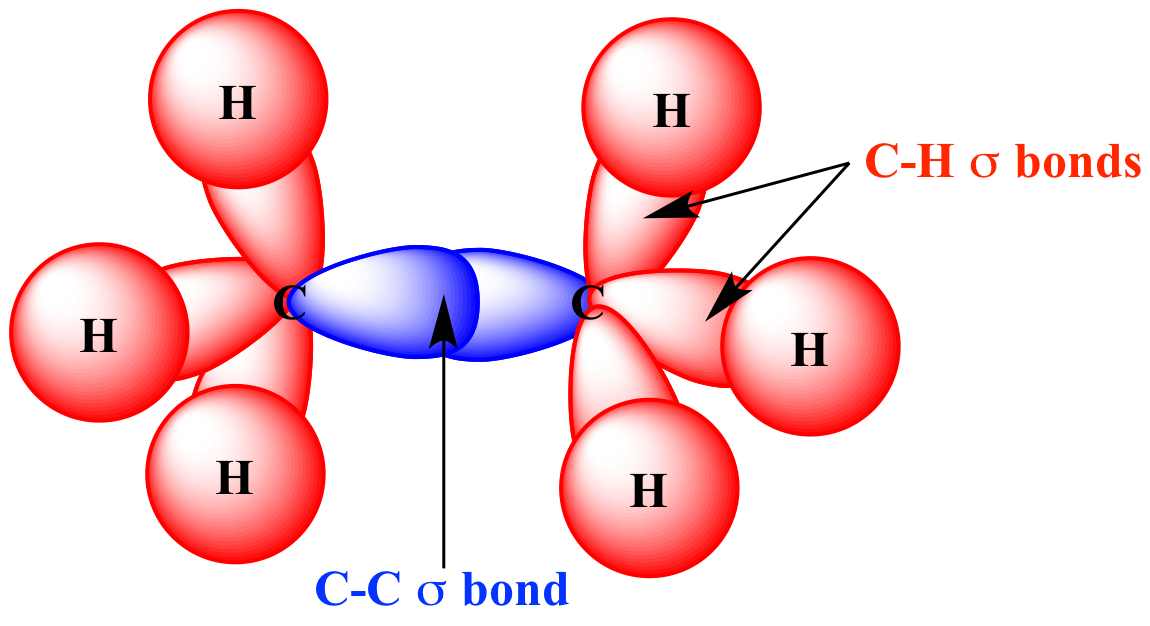
Can You Have a Pi Bond Without a Sigma Bond? These bonds can be easily calculated as Single bond= one Sigma Bond, double bond=one sigma and one pi, Triple bond= one sigma, and two Pi Bonds. How Do You Count Pi Bonds and Sigma Bonds? It depends upon the type of bonds like a single bond has one Sigma Bond, the double bond has one sigma and one Pi Bond, and the triple bond has one sigma and two Pi Bonds. Which Bond Is Stronger, Sigma or Pi Bond?Ī Sigma Bond is stronger than a Pi Bond because it has higher stability. Pi Bonds may form a double and triple bond but do not form a single bond in most cases. Sigma Bond FAQs How Are Sigma and Pi Bonds Formed?Ī Sigma Bond is formed by the face-to-face overlapping of atomic orbitals, while a Pi Bond is formed by the side-by-side overlapping.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed

